About Us
Pioneering Innovative Medical Technology Since 2006
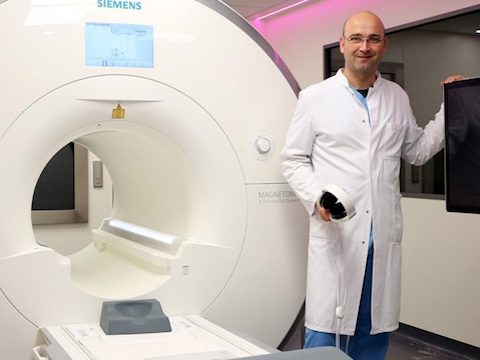
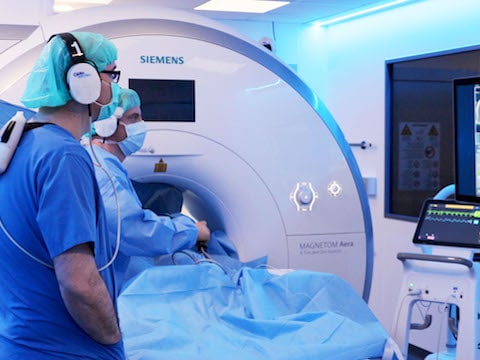
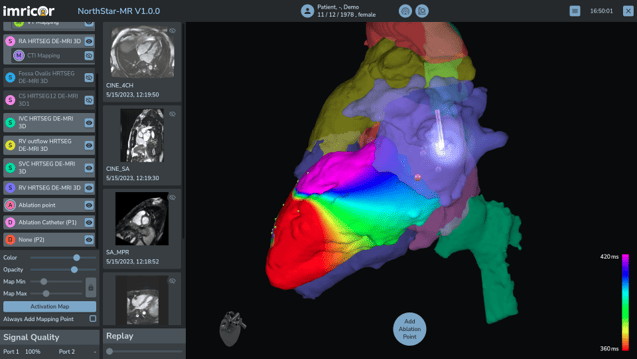
Imricor was co-founded by Steve Wedan in 2006 and along with three Imricor pioneers established in the Twin Cities area. Imricor has quickly grown to be the first and only company in the world to bring commercially viable and safe iCMR products to the catheter ablation market. As Imricor continues to grow and develop this market, its focus is and remains the improvement of ablation therapy for patients all around the world.
Receipt of CE mark approval marks a major accomplishment for the Imricor team, as we pursue our mission to improve the lives of people worldwide by delivering devices that enable iCMR procedures.
Imricor presence around the world
Board of Directors

Steve Wedan
President and Chief Executive Officer, and Board Chair
Joined May 2006

Mark Tibbles

Peter McGregor

Anita Messal

Jeffrey Leighton

Aldo Denti
Management

Steve Wedan

Jonathon Gut

Gregg Stenzel

Jennifer Weisz

Nick Corkill

Nick Twohy

Kate Lindborg

Vic Fabano