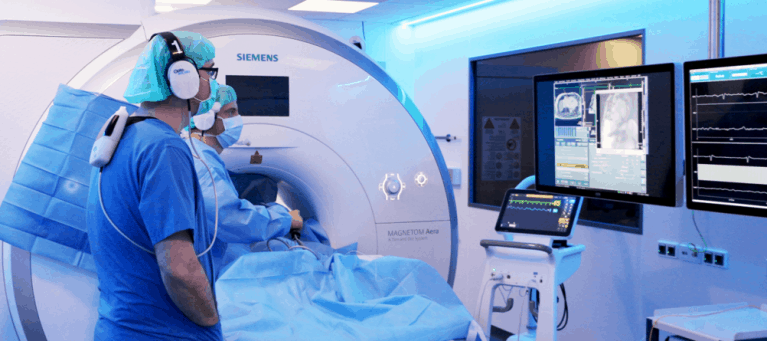
Drs. Christopher Piorkowski (left) and Thomas Gaspar (right) performing iCMR ablations at the Heart Center Dresden
05 February 2020 – Minneapolis, United States – Imricor Medical Systems, Inc. (Company or Imricor) (ASX:IMR) is pleased to announce the first procedures using the Company’s products following CE mark of the Vision-MR Ablation Catheter have been performed at the Heart Center Dresden.
Three procedures were successfully performed over two days by Dr. Christopher Piorkowski and Dr. Thomas Gaspar (https://www.herzzentrum-dresden.com/rhythmologie/unser-team/). Additional physicians at the Heart Center Dresden will also begin performing procedures immediately.
These procedures mark the first iCMR ablations anywhere in the world to ever be performed outside of a clinical trial. They were performed in an iCMR lab fitted with a Siemens 1.5T MAGNETOM Aera MR scanner.
“It’s another tremendous milestone for Imricor,” said Steve Wedan, Imricor’s Chair and CEO. “It marks the beginning of routine clinical use and the coming of age for this exciting new field of iCMR.”
Active Catheter Imaging
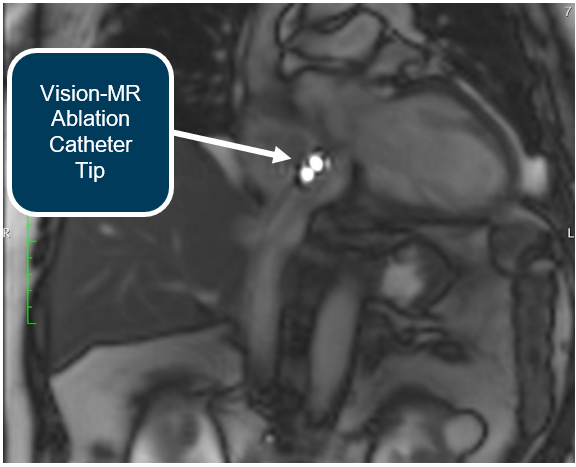
Procedures were performed, using native MR imaging on the Siemens scanner, which the Company calls Active Catheter Imaging. With this imaging technique, one can easily identify the Vision-MR Ablation Catheter without the use of active tracking or a mapping system.
Active Catheter Imaging proved to be highly effective in the procedures undertaken at Heart Center Dresden. Dr. Christopher Piorkowski commented during a procedure, “This is beautiful. It is better than fluoroscopy. In fluoroscopy you can only imagine the anatomy. Here you see it.”
Nick Twohy, Director of Marketing stated, “To complete our first cases is very exciting. We were ready to begin immediately after receiving approval. In addition to being commercially available, the Active Catheter Imaging technique enables healthcare professionals to provide care to patients today while at the same time preparing for advancements in 3D mapping technology on the horizon.”