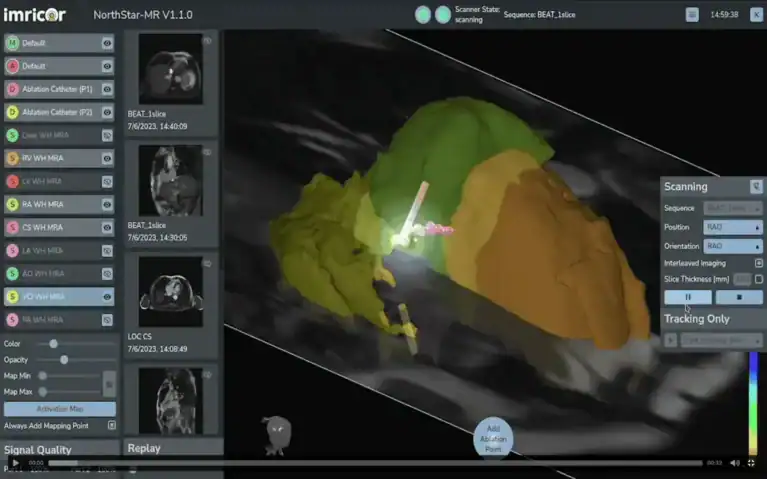
2 February 2026 – Minneapolis, MN, Imricor Medical Systems, Inc. (Company or Imricor) (ASX: IMR) is pleased to announce that the United States Food and Drug Administration (FDA) has granted 510(k) clearance for the Company’s NorthStar® Mapping System under the premarket notification process.
NorthStar is the first and only MRI-Native 3D mapping and guidance system to receive FDA clearance, setting a new standard for real-time, radiation-free visualization in cardiac electrophysiology procedures. By operating within the MRI environment, NorthStar enables physicians to use the full power of the MRI by providing real-time visibility of the anatomy, catheters, and lesion formation.
NorthStar’s clearance by the U.S. FDA marks the culmination of years of research, development, 3rd party partnerships, and regulatory work.
FDA clearance enables Imricor to commercially market NorthStar in the United States, the world’s largest electrophysiology market. This clearance is the second of what the Company expects will be multiple regulatory milestones this calendar year, as Imricor’s full MRI-guided electrophysiology platform is progressively introduced to the market.
Imricor’s VP of Marketing and Business Development, Nick Twohy, added:
“For years, our team and clinical partners have shared a vision for electrophysiology procedures that fully leverage MRI, where you can see the anatomy, your catheters, and lesion formation in real time, without any radiation. NorthStar is a major step toward making that vision routine clinical practice.
“With NorthStar at the center of the iCMR lab, physicians can perform diagnostic EP studies and ablations with the level of soft tissue detail MRI provides, while working in a familiar 3D mapping environment. Our aim is to give clinicians tools that feel intuitive on day one, yet meaningfully enhance precision, confidence, and workflow in the lab.
“Looking ahead, NorthStar as a software platform will allow us to evolve with physicians’ needs and emerging treatment strategies over time.”